It's not too late: Clinical trial seeking participants with chronic Thyroid Eye Disease (TED)
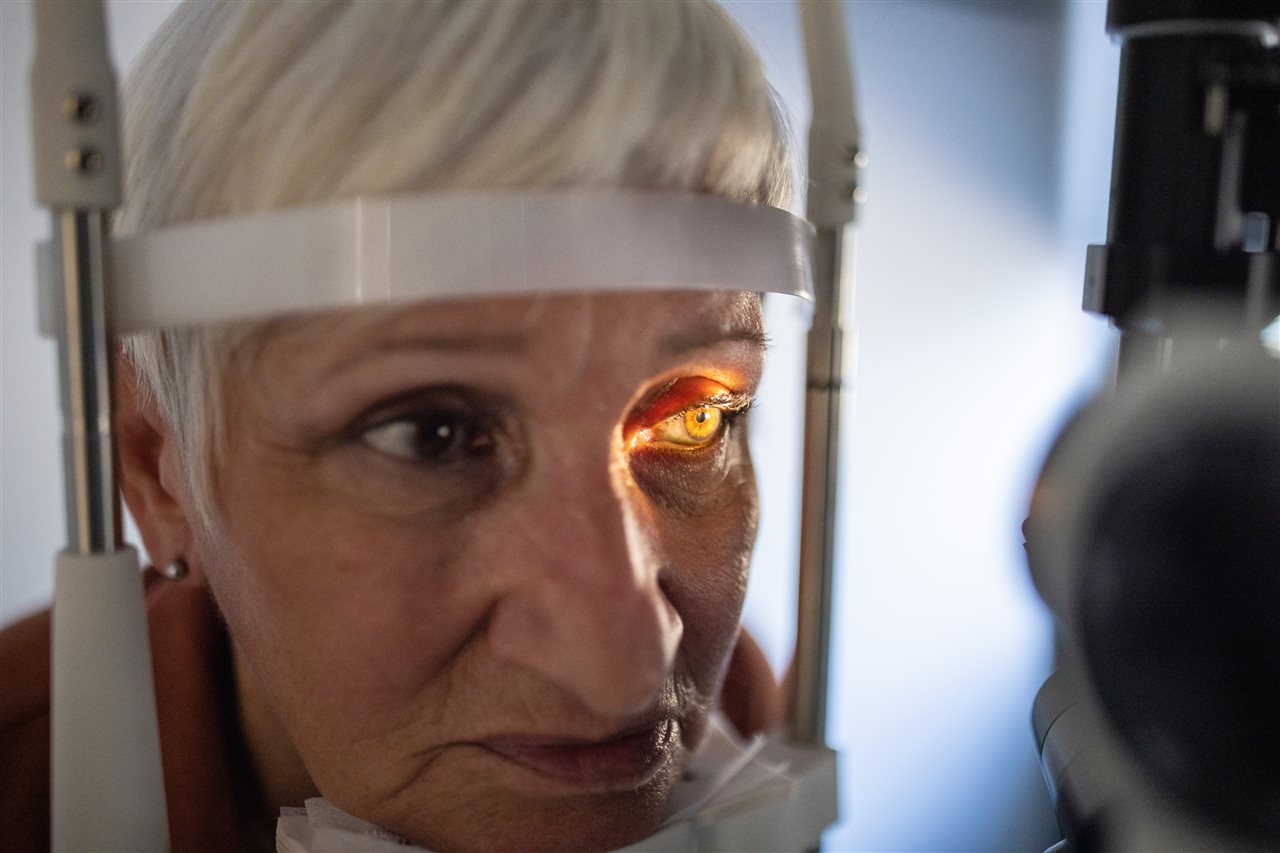
(BPT) - Imagine living for years, sometimes decades, with debilitating eye symptoms that not only affect your day-to-day activities, but often get written off or misdiagnosed by doctors. This is the unfortunate reality for many people living with a rare autoimmune condition called Thyroid Eye Disease (TED).
With TED, the immune system mistakenly attacks the muscle and fat tissue behind the eyes, causing inflammation and scar tissue to form. This can lead to bothersome and incapacitating eye symptoms like pain and bulging, double vision, redness and irritation and more.
TED is commonly experienced in two phases: the acute or active phase, when symptoms come on suddenly and may get worse quickly, followed by the chronic or inactive phase 1-3 years later, when symptoms feel more stable and less changing. But despite periods of stability, TED and its symptoms may never fully go away. As is common with autoimmune conditions, TED symptoms can flare, or come back, at any point, even in the chronic phase.
Treatment for TED
Until recently, steroids and invasive surgery were the standard of care for treating TED. Fortunately, additional options have become available to help treat TED, leading to a reduction in many symptoms, including eye bulging and double vision; but more research is needed for people who have had TED for longer than two years.
Chronic TED Study
Researchers are looking for more patients to participate in a trial to assess the safety and benefits of teprotumumab-trbw, an already FDA-approved treatment, in people with chronic (inactive) TED. Clinical trials, also known as research studies, help researchers determine how safe and effective treatments are for certain diseases.
If you live with TED, ask your doctor about enrolling in a clinical trial. To learn more about the study, and to see if you may qualify to participate, please visit www.ChronicTEDStudy.com
